|
Fat emulsion should comprise no more than 60% of the patient's total caloric intake, with carbohydrates and amino acids comprising the remaining 40% or more of caloric intake. Total Parenteral nutrition: As a part of TPN, administer IV via a peripheral vein or by central venous catheter. Impaired renal function where a concentrated source of energy may be indicated to reduce protein breakdown Ĭachexia and Patients with essential fatty acid deficiency who cannot maintain or restore a normal essential fatty acid pattern by oral intake. following cranial trauma or poisoning in cases where enteral feeding is inappropriate or impossible Nutritional disorders or disturbances of nitrogen balance due to inadequate or failing intestinal absorption caused by tumours in the gastrointestinal tract, acute or chronic intestinal diseases (peritonitis, ulcerative colitis, terminal ileitis) īurns, to reduce the frequently excessive nitrogen losses Prolonged unconsciousness, eg. Preoperative and postoperative nutritional disturbances where an improved nitrogen balance is required Part of the intravenous diet in all parenteral nutrition indications including: It is a source of essential fatty acids when a deficiency occurs. If pharmacy is filtering, what pore size filter is being used? To reduce microbial contamination, I would strictly enforce the policy of NOTHING being injected or piggybacked into this system and the system would ONLY be taken apart when it is time to change the entire administration system once every 24 hours.This is a source of calories and essential fatty acids for patients requiring parenteral nutrition for extended periods of time (usually for >5 days). To make the clinical decision about filters, I would want to know what filtration, if any, was done by the pharmacy during the compounding process. This could be considered the same as infusing a 3-in-1 PN solution thru a 1.2-micron filter. 
The other option would be to use a 1.2-micron filter in the system and infuse both 2-in-1 PN and IVFE thru the same filter. To follow these new IVFE manufacturer instructions, 2 filters of 2 different sizes must be used - a 1.2-micron filter on the IVFE and a 0.22 micron filter on the 2-in-1 PN. According to 2016 INS Standards of Practice and a 2014 ASPEN guidelines document, a 0.22-micron filter is needed for the 2-in-1 PN solution due to particulates from the compounding process and microorganisms that could enter the system. There are several options for configuring the filters. Now the IVFE requires a 1.2-micron filter regardless of where it is infused. IVFE was piggybacked below the filter or given through a separate catheter with no filtration. What about filtration for a 2-in-1 solution containing protein, carbohydrates, and other additives? The practice has been to use a 0.2-micron filter on this solution to ensure removal of all particulate matter and microorganisms. The concern was and still is cracking the fat emulsion, possibly resulting in a fat embolism. IVFE will not pass through filters with a smaller pore size. When all components are admixed together in one solution container (ie, 3-in-1 or Total Nutrient Admixture), the recommendation has always been to use an in-line 1.2-micron filter for infusion. This brings up several questions about the most appropriate filtration for infusing all parenteral nutrition solutions.  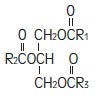
Previous instructions were not as specific, stating that filters were not recommended or that a filter of less than 1.2-micron pore size was not to be used. fat emulsion (e.g., Intralipid, Nutrilipid) states to use a 1.2-micron filter for all infusion.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
